Abstract Recently, scientists from Rice University and Moscow published a study in the American Chemical Society journal NanoLetter, using the pressureless method to try to develop ultra-thin diamond films. The staff found that under certain conditions, chemically induced phase transitions can be used for complete growth...
Recently, scientists from the University of Rice and Moscow in the United States published a study in the American Chemical Society journal Nano Letter, using the pressureless method to try to develop ultra-thin diamond film. The staff found that the chemically induced phase change method can be used to completely grow the diamond film under certain conditions without any pressure conditions and equipment. This diamond super film called dimane possesses all the superior properties of diamond such as semiconductor properties and thermal conductivity.
Diamane diamond film has broad application prospects. Sorokin, a senior researcher at Moscow's superhard materials and new carbon materials technology, said: in nanocapacitors, dimane diamond film can be used as ultra-thin dielectric hard film; used as nano in nanoelectronic devices. Parts. In addition, the nanodiabetic field is also inseparable from this superior dimane film.
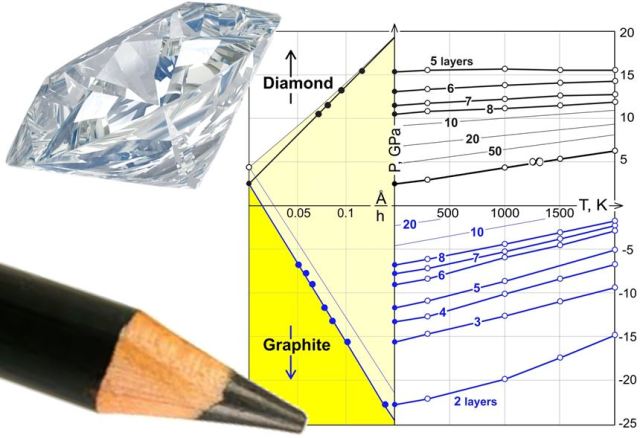
The picture shows a phase diagram of a chemically fabricated diamond film jointly developed by scientists at Rice University and Moscow. The dimane film is developed from monoatomic layer graphene.
The phase diagram of the growth of this dimane diamond film is given by the temperature, pressure and other influencing factors of the film growth, showing a complete process of transforming the graphene layer into a flawless perfect diamond crystal.
The two-dimensional material of Diamane film can not be produced by traditional methods. Scientist Richard Feynman used the concept of micro-initiation and successfully developed graphene by chemically induced phase change method of surface hydrogenation. The researchers used computer modeling to mimic the atomic force of each atom in the growth of the dimane film, including graphene atoms and hydrogen atoms (hydrogen used for catalytic reactions in experiments). It has been found that by using chemical methods, graphene is converted to diamond in the case where the experimental conditions satisfy various requirements.
The phase diagram shows the stage of influence on the ground state at each pressure point and temperature point. Yakobson added that the phase diagram for the product produced by diamane is slightly special because the final result depends on the graphene layer. The thickness and number of layers, etc., the relevant experimental parameters are all new data.
The staff also used hydrogen to do the experiment. When the catalyst hydrogen is intervened, hydrogen will carry away an electron of the graphene carbon atom, such that a bond is destroyed, and the remaining electron is left on one side of the graphene layer; The carbon atoms on the graphene sheets are very easy to bond, and the required bonding pressure is almost zero.
If the graphene layer has more than one layer but several layers, then the reaction will have a domino effect. The hydrogen of the catalyst starts from the top and then goes down. When the entire graphene layer is reacted, the complete phase change diagram comes out, and the crystal structure at this time is the diamond film.
According to Yakobson, diamond films can also be developed by CVD, but due to the characteristics of their polycrystals, CVD diamonds have certain quality defects in certain applications. Especially like some advanced electronic devices, advanced wideband gap semiconductor materials like Diamane are needed. (Compiled from 'Perfect Sheets of Diamond May be Possible Without any Pressure')
Graphite Electrodes mainly use needle coke, petroleum coke, as raw material, coal pitch as binder, calcination, ingredients, kneading, molding, baking and graphitization, machining and made, which is released in electric arc furnace in the form of arc conductor of electricity to heat melting furnace charge, Graphite Electrode with low resistivity, graphite electrode good conductivity, graphite electrode low ash, graphite electrode uniform and compact structure, good oxidation resistance, high mechanical strength, etc.
Hp 600 Graphite Electrode,Electrode Graphite For Eaf,Small Diameter Graphite Electrode,Electrodes Hp 600
Carbographite Industrial PTE.LTD , http://www.carbographite.com
